A once impossible cure for deafness is coming, thanks to gene therapy.
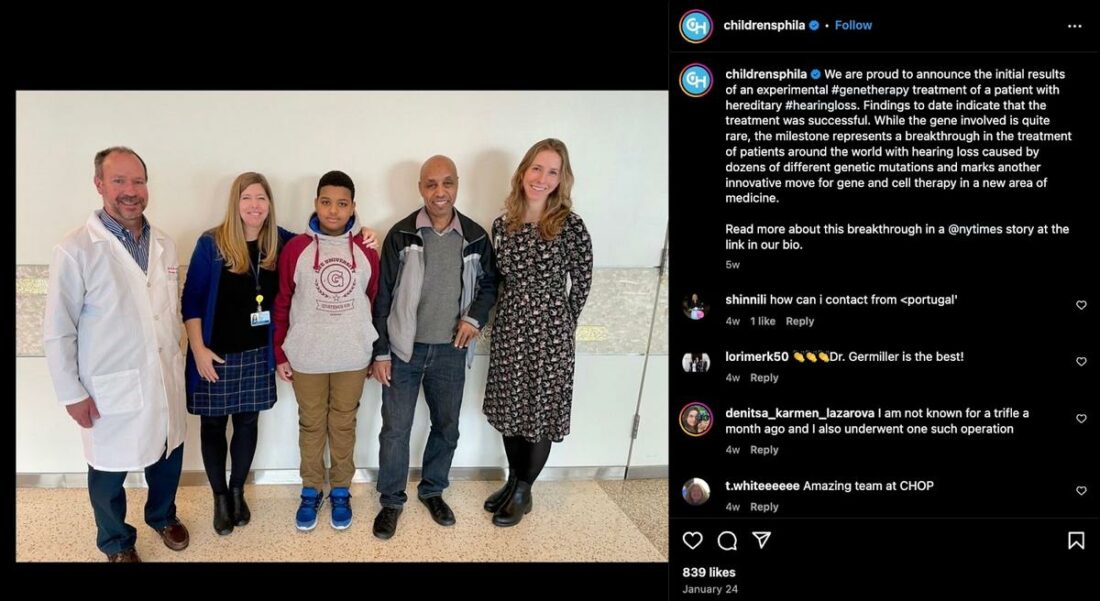
Aissam Dam, an 11-year-old boy who was born deaf due to a genetic issue, has heard for the first time as a result of a gene therapy experiment headed by Akouos Inc, part of Eli Lilly & Co.
Hearing loss, especially those you’re born with, has always been known as irreversible and used to only be temporarily assisted by hearing aids and cochlear implants. But, the breakthrough in gene therapy has challenged this belief, offering hope for millions suffering from hearing loss.
Aissam Dam’s Journey: From Silence to Sound

Imagine living in silence and then one day starting to hear the world around you. That’s what happened to Aissam Dam, an 11-year-old boy, thanks to gene therapy.
Born unable to hear because of a gene that didn’t work right, Aissam lived in a quiet world for 11 years. Then, in October 2023, he received a special treatment that was about to turn his silent world into one filled with sounds.
This treatment was developed by a company called Akouos Inc., which is part of Eli Lilly & Co. They used a new kind of therapy that fixed the gene causing Aissam’s hearing loss.
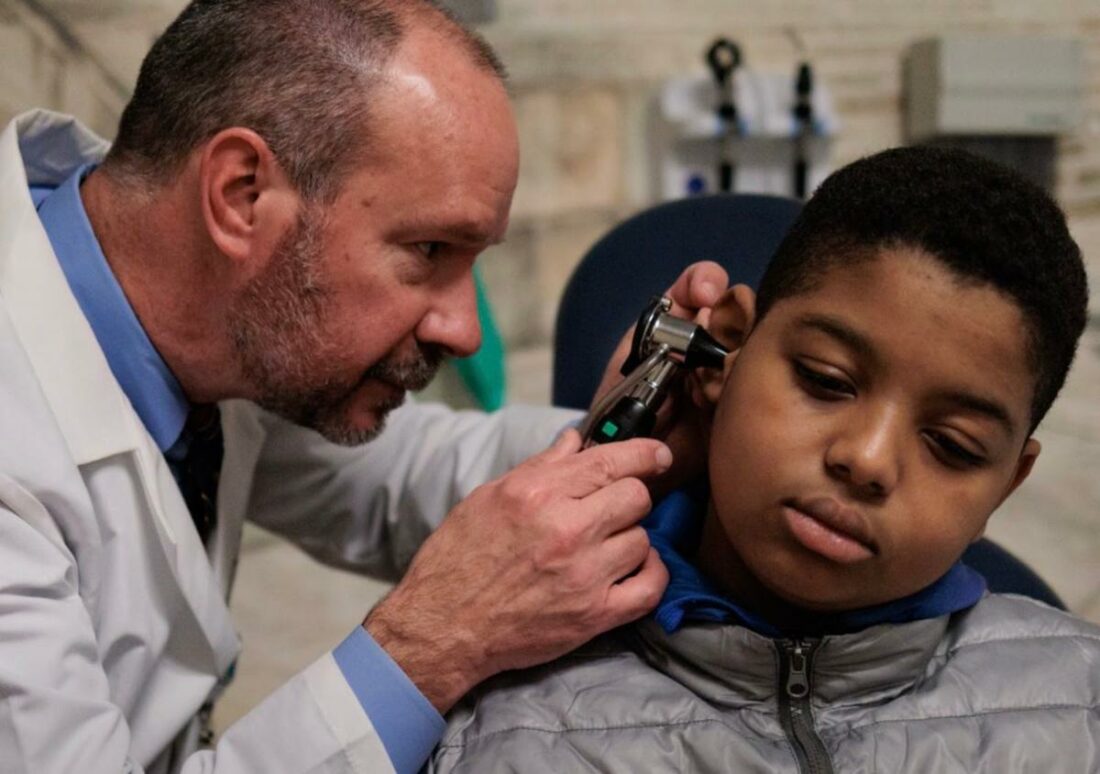
Just four weeks after getting this therapy, Aissam started to hear. For the first time, he heard cars buzzing by, the sound of his dad’s voice, and the snip of scissors during his haircut.
Aissam himself loves every new sound he discovers.
The Science of Gene Therapy
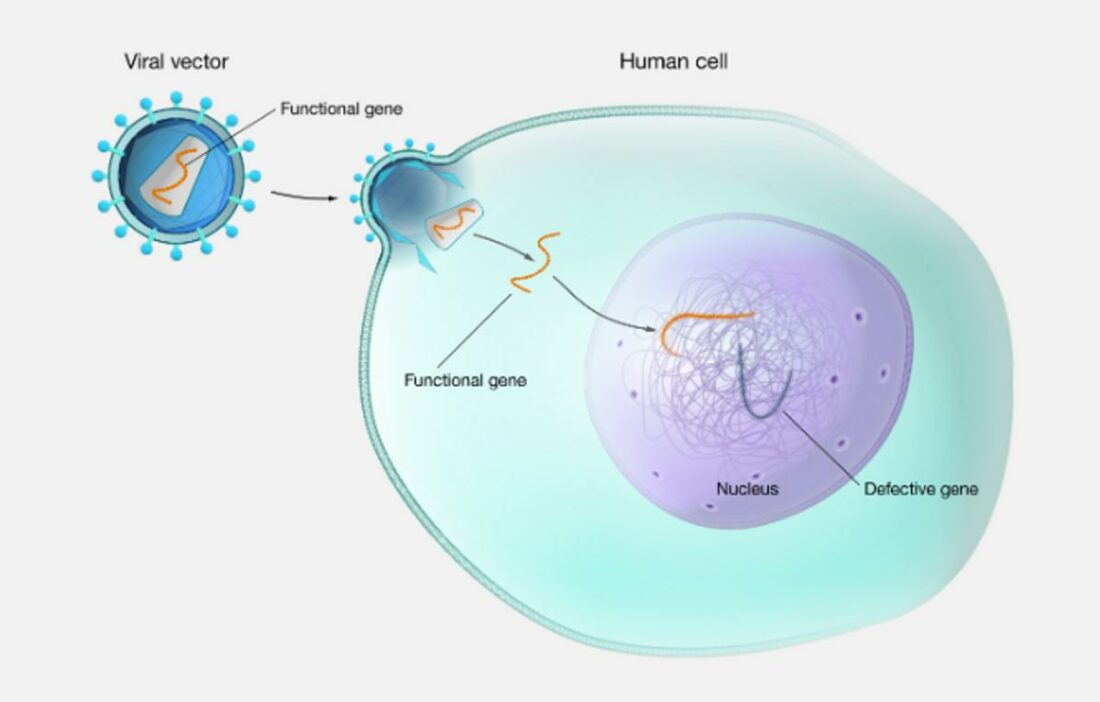
Gene therapy is like a sci-fi solution to real-life problems. It’s a cutting-edge way to treat diseases by going straight to the root: our genes.
This method either adds new genes where there are broken ones or swaps out bad genes for good ones. It’s not just for inherited diseases like hemophilia, but also for conditions that develop later, like certain kinds of cancer.

Dr. John Germiller from the Children’s Hospital of Philadelphia, who gave the gene therapy to Aissam, shared his excitement about the progress.
However, gene therapy isn’t one-size-fits-all; it’s tailored to each specific problem. In the case of hearing loss, it targets the exact genetic mutation causing the issue.
Aissam’s success story is a prime example. He had a problem with a gene that didn’t let his ears transmit sound signals properly. The therapy he got put a working version of this gene into his inner ear, attacking the problem right at its genetic source.
Other Successful Trials
The story of Aissam Dam is just one of many breakthroughs in gene therapy for hearing loss.
In Fudan, China, scientists led by Zheng-Yi Chen from the Massachusetts Eye and Ear Infirmary and Harvard Medical School, along with a team from Fudan University’s Eye & ENT Hospital, made their own mark.
They worked with six kids, aged 1 to 7, all dealing with a mutation of the OTOF (otoferlin) gene, which plays a key role in how we hear.
Out of these kids, five saw a big improvement in their hearing over six months.

This wasn’t just good news; it was life-changing. Chen captured the essence of their success, saying:
Tackling the OTOF gene’s size was a big challenge, but the team had a smart solution. They split the gene in two, used different viruses to carry each part, and then combined them in the patient’s cells. This clever workaround not only led to successful treatments but also laid the groundwork for future therapies.

Yilai Shu, another key player in the study, shared the emotional impact of their work:
Challenges and Future Directions in Gene Therapy for Hearing Loss
Even with the exciting progress in gene therapy for hearing loss, there are significant hurdles to overcome.
Right now, these treatments are designed for very specific genetic mutations. This means they can’t yet help the vast majority of people with hearing loss.
A big challenge is the nature of genetic deafness itself. In many cases, it’s caused by the loss of cochlear hair cells, which don’t regenerate.
However, there’s good reason for hope. The successes in gene therapy trials lay the groundwork for future research, aiming to make these treatments work for a broader range of hearing loss.
Zheng-Yi Chen, a pioneer in the field, shares an optimistic view:
Looking to the future, researchers plan to keep a close eye on current study participants and start new trials with a more diverse group. With hopes of regulatory approval in the next three to five years, the path forward for gene therapy in hearing loss is promising.
